Gladstone NOW: The Campaign Join Us on the Journey✕

From studying the virus to developing tests, treatments, and therapies for long COVID, Gladstone scientists have evolved their COVID-19 research to meet critical challenges. Seen here, Senior Staff Research Scientist Ursula Schulze-Gahmen working in the lab.
When the COVID-19 pandemic began, Gladstone scientists sprung into action. Over time, our goals evolved from understanding the basic biology of the virus to creating diagnostic tests, finding new treatments, predicting new variants, and treating long COVID. Since 2020, we have:
- Published more than 100 scientific papers on COVID-19 and long COVID.
- Brought two new drugs developed at Gladstone into clinical trials.
- Launched two new companies to bring a preventative treatment and a diagnostic tool to market.
- Established more than 60 academic collaborations across the nation.
- Unlocked $85 million in grant funding through philanthropic support.
- Recruited two additional virologists, including a vaccine specialist.
None of this would have been possible without support from donors. You answered the call before the public funding agencies could, directing essential and flexible resources exactly where and when they were needed. Without your partnership, we could not have made the groundbreaking progress you’ll read about below.
Establishing a state-of-the-art-facility to study the virus.
In 2020, Gladstone brought a dedicated biosafety lab online to meet the high containment standards necessary to study COVID-19. With the added level of protection that a BSL-3 lab provides, researchers can work directly with the virus. Gladstone’s facility is exceptional because it houses multiple facilities in one, allowing scientists to eliminate extraneous steps and complete their research in one place.
Gaining a more complete understanding of long COVID.
Nadia Roan, PhD and her lab are working to understand how the immune system goes awry in long COVID. Recently, they found that individuals with long COVID have “exhausted” immune cells that are not coordinated with the antibody arm of immunity. These findings highlight a mismatch in immune system activity and suggest that long COVID may involve low-level viral persistence.

Melanie Ott speaking with a postdoc in her office.
Long COVID is challenging to study in part due to a lack of animal models of the disease. Since there is no standard diagnostic test for long COVID, it’s difficult to discern whether mice previously infected with COVID-19 experience the brain fog, fatigue, and other associated symptoms seen in humans. Nadia Roan, PhD, Melanie Ott, MD, PhD, and Jorge Palop, PhD have developed a long COVID model by repurposing an AI platform originally developed to study behavioral changes in neurological disease. The model is able to detect the hallmarks of long COVID in mice and the team has identified a potential therapeutic intervention. They’re also expanding the model in the context of long COVID risk factors such as diabetes.
Predicting new mutations.
The ability to synthesize viral genetic material for experimentation in labs is key to viral research. Due to risks associated with propagating viruses in a lab, Taha Taha, PharmD, PhD, in collaboration with Melanie Ott, MD, PhD, Jennifer Doudna, PhD, and others developed a strategy to generate noninfectious replicating particles of a virus. These particles are not pathogenic but can “stand in” for the real virus for study. Recently, the team is combining this technology in a high-throughput experimental-AI platform to predict how viruses will mutate.
Learning how COVID-19 leads to blood clots.
Research led by Katerina Akassoglou, PhD and Warner Greene, MD, PhD has revealed that the blood protein fibrin is a primary cause of the unusual blood clotting associated with COVID-19, overturning the prevailing hypothesis that it is caused by inflammation. This could also account for the neurological symptoms that occur in both COVID-19 and Long COVID, such as brain fog. A drug targeting this toxic clotting is already in Phase 1 clinical trials.
Designing a better COVID-19 test.
Melanie Ott, MD, PhD, Jennifer Doudna, PhD, and Katie Pollard, PhD have developed a new type of COVID-19 test that uses CRISPR technology. The diagnostic combines the accuracy of a PCR test with the low cost, portability, and simplicity of an antigen test. Not only can the test detect whether a sample is positive for the COVID-19 virus, it can also quantify how much virus is there and distinguish between COVID-19, RSV, and the flu in a single sample. Scientists are now planning to repurpose the test for HIV and potentially other viruses such as dengue.
The team has now launched a new company, DirectBio, to advance CRISPR diagnostics to the clinic. They have also built a Global Scholar Program to train scientists and doctors from the global south in building, using, and adapting CRISPR diagnostics.
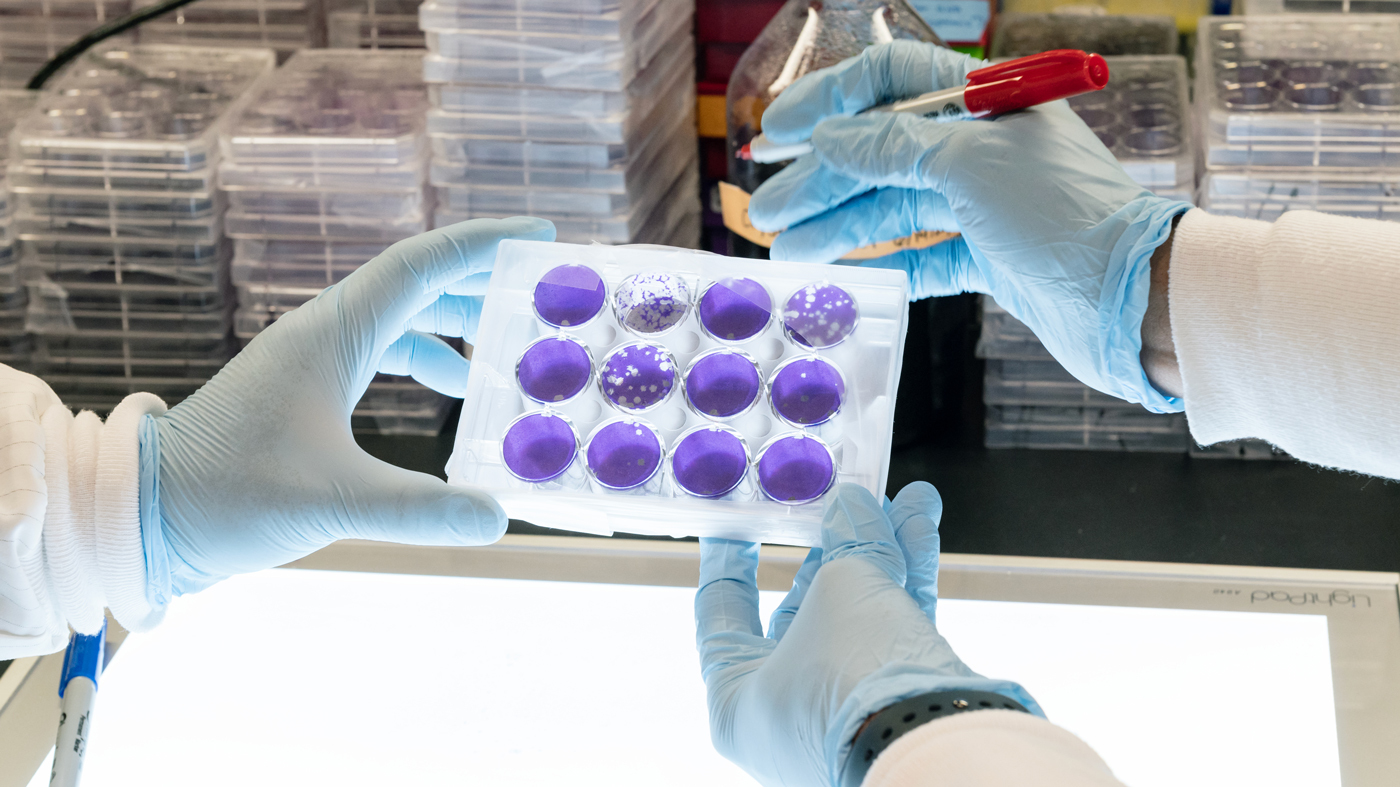
Scientist in the Ott Lab, looking at plaque assay plates.
Mapping new drug targets.
Nevan Krogan, PhD, and his team have mapped the points of interaction between our cells and the virus. They have identified 66 druggable human proteins that are essential for viral entry and replication. These proteins are targeted by 69 compounds, of which 29 are already FDA-approved, 12 are in clinical trials, and 29 are preclinical compounds. One of the drugs identified through this approach is currently in clinical trials.
Stopping viral spread.
A team led by Warner Greene, MD, PhD discovered a new way to prevent COVID-19 transmission. Repurposing research originally done on HIV, they developed a nasal spray that targets the spike protein and blocks the viral fusion necessary for the virus to enter our cells. A new company, Invisishield Technologies, was launched to bring this novel preventive treatment to the public.
Modeling lung function to study disease.
The Ott Lab has developed human lung organoids to serve as accurate and analogous models of human lungs to study how COVID-19 affects our airways. Using these organoids, scientists have studied the impact of vaccination on immunity, differences between variants, mechanism of action of new antivirals, and more. The team has filed patent applications for two antivirals tested using this system and the BSL-3.
Understanding implications for the heart.
While COVID-19 is primarily a respiratory disease, it can also cause heart damage that lingers even after recovery. Gladstone scientist Bruce Conklin, MD, and his team studied how COVID-19 damages the heart at the cellular level.
They found that the virus damages the heart’s muscle cells, which are necessary for the contraction and relaxation involved in heartbeats. Many heart cells exhibited missing nuclear DNA. This damage was observed irrespective of disease severity, such that even patients with mild disease might suffer lasting heart damage.

Melanie Ott alongside attendees at the Industry Partnership Forum.
Examining immune responses to COVID-19.
Nadia Roan, PhD and her team examined different immune responses to COVID-19, unearthing clues to what makes some people’s immune systems better or worse at fighting COVID. They also found ways to predict immune response and compared immunity from vaccines to immunity from prior infection.
Featured Experts
Support Discovery Science
Your gift to Gladstone will allow our researchers to pursue high-quality science, focus on disease, and train the next generation of scientific thought leaders.







