Gladstone NOW: The Campaign Join Us on the Journey✕

Mauricio Montano will serve as the BSL-3 lab director.
In the race to develop treatments and vaccines for SARS-CoV-2, the virus causing COVID-19, scientists need to be able to study the virus. However, the virus’s high degree of infectivity puts scientists at high-risk when they work with the active virus. In order to advance toward effective diagnostics, treatments, and vaccines for a disease with more than 14 million documented cases globally, scientists need a safe and effective way to handle this dangerous pathogen in a controlled environment. A Biosafety Level 3 (BSL-3) lab is designed to meet these special requirements.
Many scientists and labs are shifting focus to studying SARS-CoV-2, bringing the relative scarcity of BSL-3 labs into sharp focus. Respiratory viruses, such as SARS-CoV-2, require a special type of BSL-3 containment lab facility designated for airborne pathogens. To achieve this designation level, labs must meet technical standards that can be costly and demand significant obligations to implement. A voluntary reporting of BSL-3 labs in 2018 revealed that there are just over 200 in the United States. The Bay Area has a handful of them, including on the campuses of UC Berkeley and the Lawrence Livermore National Laboratory. Now, Gladstone Institutes, located in the Mission Bay neighborhood of San Francisco, California, is home to an airborne pathogen BSL-3 lab as well. Gladstone faces a unique opportunity to leverage its expertise in virology and emerging infectious diseases and the availability of this facility will allow its scientists to directly address the COVID-19 pandemic.
Fortunately, Gladstone didn’t need to start from scratch to bring their BSL-3 lab online. An existing containment lab meeting some BSL-3 specifications already existed, giving Gladstone a head start compared to building a functional facility from the ground up, which can sometimes take years. In contrast, Gladstone’s timeline has been only a few months. In that time, the air filtration system has been updated, new lab equipment has been brought in and tested, procedures for safely operating within the lab have been detailed, and specialized training of lab personnel has been conducted. Amid a sharp increase in global demand, critical personal protective equipment (PPE) suited for this special environment was also procured.
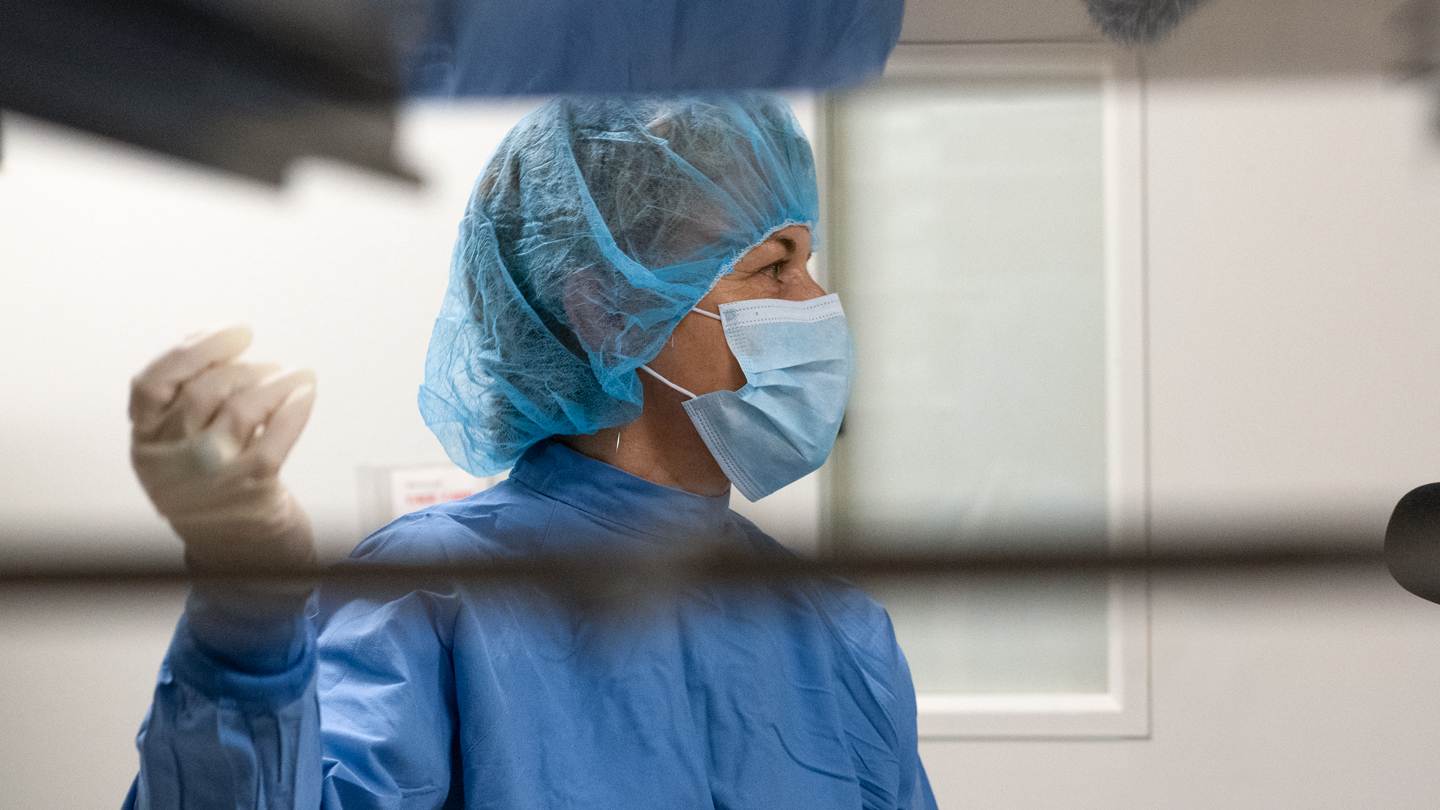
The certification and commissioning of this BSL-3 facility required following strict physical and procedural guidelines. The lab is secured and sits behind a set of alarmed and self-closing, locking doors that connect to the rest of the building through a hallway. The hallway itself acts as a barrier between the lab doors and the general corridors leading to the rest of the building. Airflow is carefully controlled to prevent recirculation by drawing fresh air in from outside and strict air filtration is in place. Access to the lab is restricted to highly trained lab personnel. Gladstone staff, whether researchers or maintenance and facilities crews, are barred from entering without first completing the extensive training. And, anyone working in the lab must be authorized and wear the highest level of PPE at all times, including respirators, closed front smocks, and eye protection.
These features are what make the BSL-3 lab challenging and expensive for a research organization to maintain. Most biomedical research is performed in labs with less restricted environments typically designated as BSL-1 or BSL-2 levels. But for more dangerous pathogens, additional levels of protection are necessary.
“In a BSL-2 lab, there’s only so much you can learn from inactive parts of the SARS-CoV-2 virus,” says Gladstone BSL-3 Lab Director Mauricio Montano. “You’re finding answers here and there, but they don’t necessarily give a full picture of how the live virus will react in a given situation.”
For example, researchers can see how the virus as a whole reacts to different drugs, and identify candidate therapeutics by finding the drugs that most effectively block the virus from spreading.
Melanie Ott, MD, PhD, director of the Gladstone Institute of Virology, spearheaded Gladstone’s effort to refurbish its BSL-3 lab, and will use it to test host:virus interactions in different target cells and the effect of FDA-approved drugs on SARS-CoV-2. If an already approved drug can interrupt the virus’s interaction with human cells, it can be studied further as a potential therapy in treating COVID-19.
Melanie Ott spearheaded the effort to refurbish Gladstone's BSL-3 facility.
“I am thankful to the many supporters both internally and externally that have supported our effort to develop this facility,” says Ott. “Our efforts to combat this virus will be aided significantly by the BSL-3 opening. It will be exciting to see what additional impact Gladstone and our collaborators will make in the upcoming months.”
The lab is another example of Gladstone’s commitment to evolving relentlessly and a testament to embracing change. Gladstone President Deepak Srivastava, MD, sees it as a natural step saying, “Our operational agility is what sets us apart from other biomedical research institutions. The refurbishing of our BSL-3 lab is just one example of our capabilities and has the potential to create impact felt globally.”
Srivastava is confident the lab will go beyond finding breakthroughs for SARS-CoV-2 and envisions a future where some of the most complicated diseases will be studied within Gladstone’s walls. “We’re fortunate to have brilliant minds throughout our organization who will ensure the lab fulfills its highest potential.”
Montano and Ott recently addressed questions about the opening of the new facility at Gladstone.
For Media
Julie Langelier
Associate Director, Communications
415.734.5000
Email
About Gladstone Institutes
Gladstone Institutes is an independent, nonprofit life science research organization that uses visionary science and technology to overcome disease. Established in 1979, it is located in the epicenter of biomedical and technological innovation, in the Mission Bay neighborhood of San Francisco. Gladstone has created a research model that disrupts how science is done, funds big ideas, and attracts the brightest minds.
Support Our COVID-19 Research Efforts
Gladstone scientists are moving quickly to respond to the coronavirus outbreak. Help us end this pandemic.
Gladstone Recognized in Inaugural Cure Innovation Index for Translating Science Into Real-World Health Impact
Gladstone Recognized in Inaugural Cure Innovation Index for Translating Science Into Real-World Health Impact
Gladstone ranks 15th among 60 institutes and centers nationally for its ability to turn discoveries into measurable health outcomes.
Institutional News News ReleaseGladstone Expands Research Footprint With 105,000 Square Feet of New Laboratory Space
Gladstone Expands Research Footprint With 105,000 Square Feet of New Laboratory Space
Gladstone Institutes is adding state-of-the-art laboratories in a newly constructed Mission Bay building.
History Institutional News News Release Genomic Immunology Data Science and Biotechnology Marson Lab Pollard Lab AI CRISPR/Gene Editing GenomicsOne Person’s Final Gift to Science Gets Us Closer to an HIV Cure
One Person’s Final Gift to Science Gets Us Closer to an HIV Cure
A new documentary follows Jim Dunn’s end-of-life decision to donate his tissues to HIV research.
Institutional News HIV/AIDS Infectious Disease Roan Lab





