Gladstone NOW: The Campaign Join Us on the Journey✕

Mark Petersen (left) and his team—including Caroline Brandt (right)—identified a root cause of developmental delays that result from brain bleeds in premature infants, opening a path for new therapeutic strategies to counter long-term health risks.
Mark Petersen, MD, has seen firsthand the devastating effects of brain bleeds in premature babies. It’s an exceedingly common condition that affects up to 20 percent of infants born before 28 weeks of gestation, bringing an increased risk for developmental delays and autism.
“As a neonatologist and neuroscientist, it’s frustrating that we don’t have any treatments to counteract the harmful effects of bleeding in the developing brain, even though we know it often leads to lasting problems,” says Petersen, director of the Neuro-Intensive Care Nursery at UC San Francisco (UCSF), associate professor of pediatrics at UCSF, and a visiting scientist at Gladstone Institutes. “Adding to this frustration, we’ve had very little understanding—until now—of why and how this bleeding is so closely tied to the long-term neurological issues these babies often face.”
In a study that appears in Proceedings of the National Academy of Sciences (PNAS), Petersen and an interdisciplinary team of physicians and scientists from Gladstone and UCSF shed light on this vexing medical condition, showing for the first time that a blood protein called fibrin blocks an essential biological process that drives brain development in early life.
“Toxic effects of blood in the developing brain point to potential new causes for neurodevelopmental disorders associated with preterm births.”
When babies are born extremely prematurely, the tiny and fragile blood vessels in their brain can break, causing a hemorrhage; the younger the baby, the higher the risk. Why this happens is not fully known, but poor neurological outcomes from brain bleeding are well established, explains Petersen, who leads his research laboratory at the UCSF Newborn Brain Research Institute.
In the new study, scientists demonstrated in mouse models that fibrin, which normally works to enable blood clotting, interferes with a cell-signaling pathway that plays a vital role in neuron creation—particularly in the cerebellum. They also confirmed their findings using brain scans from nearly 60 preterm infants. No other blood protein had the same inhibitory effect on this pathway, Petersen says, which allowed the team to pinpoint fibrin as a clear therapeutic target for treating babies who experience brain bleeds.
A Druggable Target
The study’s findings hold great significance for neonatal care, but also expand into other realms of medicine, as the signaling pathway (known as the “sonic hedgehog” pathway, or SHH) at the center of the study plays other important roles in human development and is implicated in a wide range of diseases.
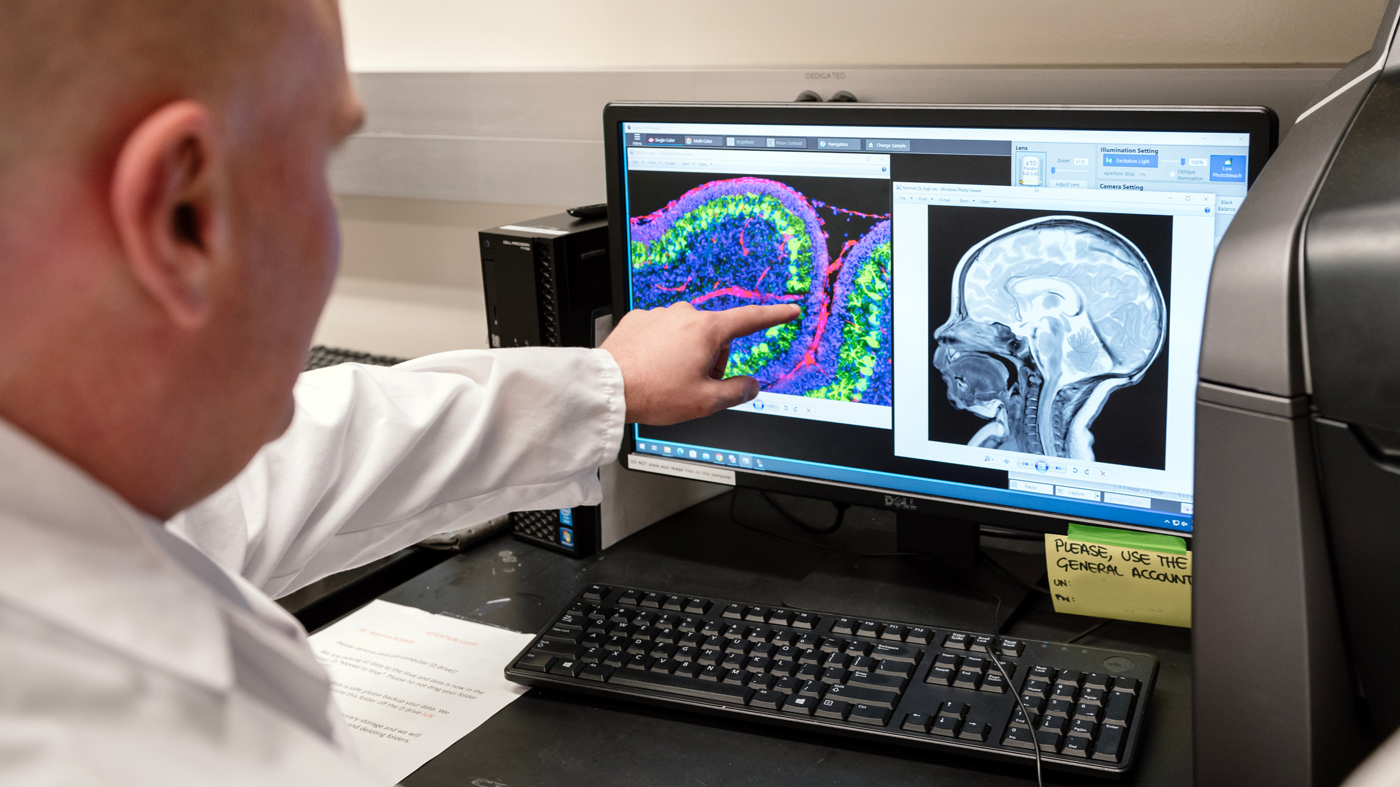
Petersen and his colleagues conducted experiments in mice to understand how the blood-clotting protein fibrin interferes with brain growth in preterm babies. The image on the left of his screen is a cerebellum of a neonatal mouse with inflammatory brain injury; fibrin appears in red. The team also analyzed MRIs from preterm infants.
At Gladstone, Petersen worked closely with the lab of Senior Investigator Katerina Akassoglou, PhD, who has long investigated how blood that leaks into the brain triggers neurologic diseases, essentially by hijacking the brain’s immune system and setting off a cascade of harmful, often-irreversible effects leading to problems with cognition and motor functions.
“Toxic effects of blood in the developing brain point to potential new causes for neurodevelopmental disorders associated with preterm births,” says Akassoglou, who also serves as director of the Gladstone-UCSF Center for Neurovascular Brain Immunology. “Neutralizing the toxic effects of fibrin in the brain presents a promising therapeutic approach for many neurological diseases—and now, we believe may also be an approach for treating the youngest patients of all.”
Akassoglou’s lab previously developed a drug, a therapeutic monoclonal antibody, that acts only on fibrin’s harmful properties without affecting its beneficial role in blood clotting. This fibrin-targeting immunotherapy protects from multiple sclerosis and Alzheimer’s disease in mice. A humanized version of this first-in-class fibrin immunotherapy is already in Phase 1 safety clinical trials by Therini Bio.
“We’re encouraged that we’ve been able to confirm the cause of lasting neurological issues in preterm babies with brain bleeding, and that we have a druggable target to potentially address this unmet need,” Petersen says. “Our new study establishes the groundwork to test our therapeutic approach in developmental brain injury and other disease models with neurovascular disruption or abnormal SHH signaling.”
Changing Life’s Trajectory
Petersen has been leading the study for the past three years, starting with observations that premature babies often have a smaller cerebellum over time. By working with MRI experts and neurologists at UCSF to follow a cohort of 59 preterm infants, the team was able to show that the smaller cerebellum was linked to brain bleeding rather than infections or other possible causes.
“The team made a really interesting discovery that may one day change the whole life trajectory of young patients with brain bleeds.”
“During this stage of early development, the cerebellum is going through a rapid change and is very sensitive to injury,” Petersen says. “Anything that blocks the SHH pathway would have major implications on the brain, and it led us to investigate whether fibrin could be at play.”
In addition to the MRI analyses—led by UCSF pediatric neurologist Dawn Gano, MD—the team conducted experiments in animal models to fully understand how fibrin impacts the SHH pathway and neuron growth.
Lennart Mucke, MD, director of the Gladstone Institute of Neurological Disease, noted that the research addresses a fundamental biomedical question and has the potential to prevent persistent neurological problems stemming from a person’s earliest days of life.
“The team made a really interesting discovery that may one day change the whole life trajectory of young patients with brain bleeds,” Mucke says. “The focus now is on bringing this work to the clinic as quicky as possible.”
For Media
Kelly Quigley
Director, Science Communications and Media Relations
415.734.2690
Email
About the Study
The study, “Fibrinogen inhibits sonic hedgehog signaling and impairs neonatal cerebellar development after blood-brain barrier disruption,” was published on July 23, 2024, in Proceedings of the National Academy of Sciences (PNAS). Authors include Olivia Weaver, Dawn Gano, Yungui Zhou, Hosung Kim, Reshmi Tognatta, Zhaoqi Yan, Jae Kyu Ryu, Caroline Brandt, Trisha Basu, Martin Grana, Belinda Cabriga, Maria del Pilar S. Alzamora, A. James Barkovich, Katerina Akassoglou, and Mark A. Petersen. The work was supported by the National Institutes of Health and the Simon Family Trust.
About Gladstone Institutes
Gladstone Institutes is an independent, nonprofit life science research organization that uses visionary science and technology to overcome disease. Established in 1979, it is located in the epicenter of biomedical and technological innovation, in the Mission Bay neighborhood of San Francisco. Gladstone has created a research model that disrupts how science is done, funds big ideas, and attracts the brightest minds.
Featured Experts
Gladstone Launches Center for PhAIge Therapy to Harness AI in the Fight Against Drug-Resistant Infections
Gladstone Launches Center for PhAIge Therapy to Harness AI in the Fight Against Drug-Resistant Infections
The center, funded by an NIH grant, will become one of three national centers dedicated to accelerating the development of phage therapy.
News Release Grants Data Science and Biotechnology Infectious Disease Shipman Lab Pollard Lab Ott Lab Silas Lab AIGladstone Investigator Andrew Yang Receives 2026 Sobrato Prize in Neuroscience
Gladstone Investigator Andrew Yang Receives 2026 Sobrato Prize in Neuroscience
The prize honors scientists whose breakthroughs in brain research have high potential for patient impact.
Philanthropy Awards News Release Alzheimer’s Disease Neurological Disease Yang LabMapping How the Brain Takes Out Its Trash
Mapping How the Brain Takes Out Its Trash
Gladstone scientists developed a new tool to trace how the brain clears “waste,” uncovering surprising new biology about how it keeps itself healthy.
News Release Research (Publication) Alzheimer’s Disease Neurological Disease Yang Lab




