Gladstone NOW: The Campaign Join Us on the Journey✕

Scientists found that without vaccination, infection with Omicron does not provide robust immunity against other COVID-19 variants. In this photo are the study's first authors, Tongcui Ma (left), Irene Chen (center), and Rahul Suryawanshi (right).
In unvaccinated people, infection with the Omicron variant of SARS-CoV-2 provides little long-term immunity against other variants, according to a new study by researchers at Gladstone Institutes and UC San Francisco (UCSF), published today in the journal Nature.
In experiments using mice and blood samples from donors who were infected with Omicron, the team found that the Omicron variant induces only a weak immune response. In vaccinated individuals, this response—while weak—helped strengthen overall protection against a variety of COVID-19 strains. In those without prior vaccination, however, the immune response failed to confer broad, robust protection against other strains.
“In the unvaccinated population, an infection with Omicron might be roughly equivalent to getting one shot of a vaccine,” says Melanie Ott, MD, PhD, director of the Gladstone Institute of Virology and co-senior author of the new work. “It confers a little bit of protection against COVID-19, but it’s not very broad.”
“This research underscores the importance of staying current with your vaccinations, even if you have previously been infected with the Omicron variant, as you are still likely vulnerable to re-infection,” says co-senior author Jennifer Doudna, PhD, who is a senior investigator at Gladstone, a professor at UC Berkeley, founder of the Innovative Genomics Institute, and an investigator of the Howard Hughes Medical Institute.

Scientists at Gladstone and UCSF detail the immune response conferred by infection from different COVID-19 variants. In this photo are two of the study's senior authors, Melanie Ott (left) and Nadia Roan (right).
A Weaker Infection
As the Omicron variant of SARS-CoV-2 spread around the globe in late 2021 and early 2022, anecdotal evidence quickly mounted that it was causing less severe symptoms than Delta and other variants of concern. However, scientists weren’t initially sure why that was, or how a weaker infection might impact long-term immunity against COVID-19.
“When the Omicron variant first emerged, a lot of people wondered whether it could essentially act as a vaccine for people who didn’t want to get vaccinated, eliciting a strong and broad-acting immune response,” says Irene Chen, co-first author of the new study and graduate student in Ott’s lab. Other first authors are Rahul Suryawanshi, PhD, a Gladstone staff research scientist, and Tongcui Ma, PhD, scientist in the Roan Lab at Gladstone.

Scientists confirmed that, despite the milder symptoms associated with Omicron, the variant still takes hold in cells and generates an immune response. Left to right: Irene Chen, Rahul Suryawanshi, and Tongcui Ma.
To find the answer, the team of researchers first examined the effect of Omicron in mice. Compared to an ancestral strain of SARS-CoV-2 and the Delta variant, Omicron led to far fewer symptoms in the mice. However, the virus was detected in airway cells, albeit at lower levels. Similarly, Omicron was able to infect isolated human cells but replicated less than other variants.
The team then characterized the immune response generated by Omicron infections. In mice infected with Omicron, despite the milder symptoms, the immune system still generated the T cells and antibodies typically seen in response to other viruses.
“We demonstrated in this study that the lower pathogenicity of Omicron is not because the virus cannot take hold,” says Nadia Roan, PhD, an associate investigator at Gladstone.
That leaves other reasons that might explain why Omicron differs from other variants in terms of symptoms and immunity, including the lower replication seen with Omicron or the types of antibodies that the immune system generates in response to the virus.
No Cross-Variant Protection
To gauge how the immune response against Omicron fared over time, the researchers collected blood samples from mice infected with the ancestral, Delta, or Omicron variants of SARS-CoV-2 and measured the ability of their immune cells and antibodies to recognize five different viral variants—ancestral (WA1), Alpha, Beta, Delta, and Omicron.
Blood from uninfected animals was unable to neutralize any of the viruses—in other words, block the ability of any of the viruses to copy themselves. Samples from WA1-infected animals could neutralize Alpha and, to a lesser degree, the Beta and Delta virus—but not Omicron. Samples from Delta-infected mice could neutralize Delta, Alpha and, to a lesser degree, the Omicron and Beta virus.
However, blood from Omicron-infected mice could only neutralize the Omicron variant.
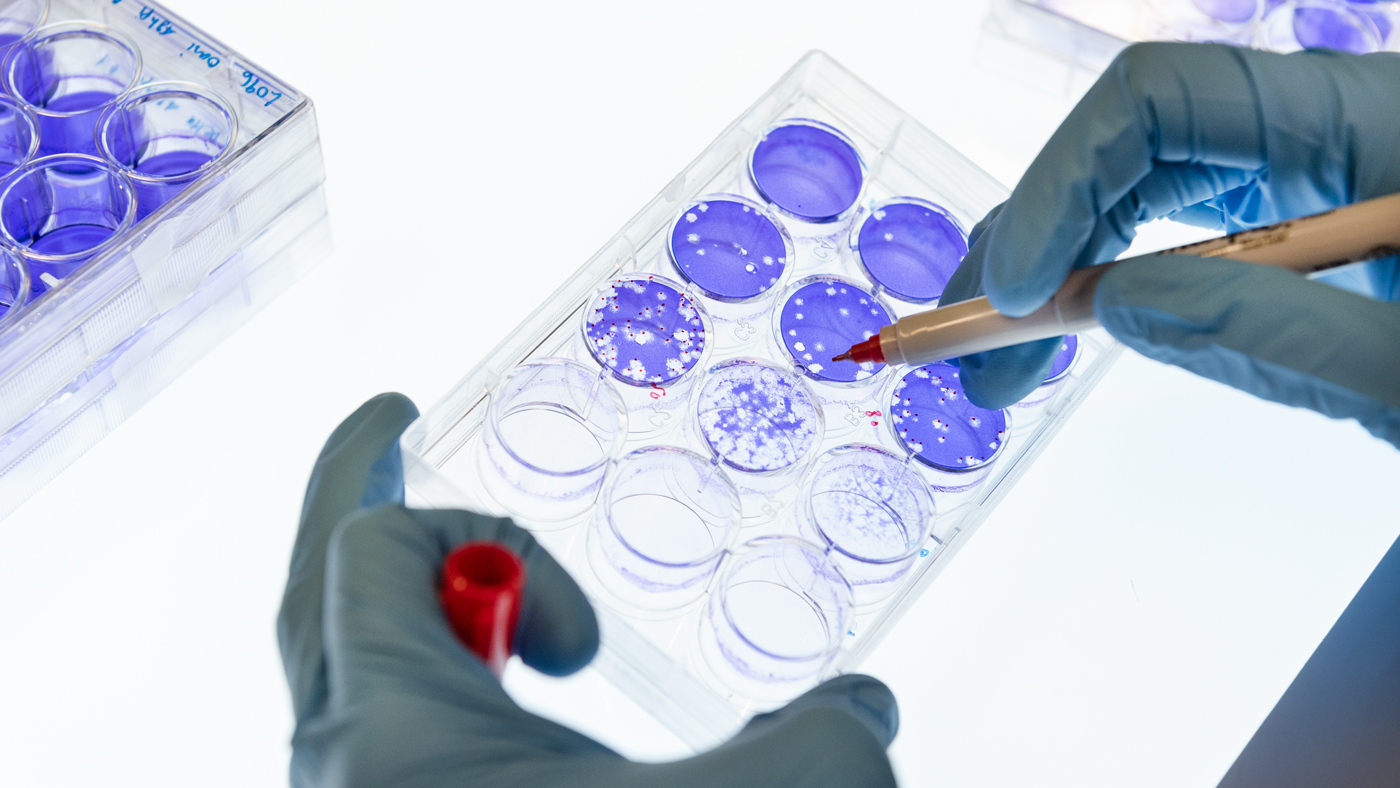
The new study shows that infection with Omicron does not protect against other variants of COVID-19. In this photo, clear zones on the purple background show the SARS-CoV-2 virus escaping from neutralizing antibodies in patient blood samples.
The team confirmed these results using blood from ten unvaccinated people who had been infected with Omicron—their blood was not able to neutralize other variants. When they tested blood from 11 unvaccinated people who had been infected with Delta, the samples could neutralize Delta and, as had been seen in mice, the other variants to a lesser extent.
When they repeated the experiments with blood from vaccinated people, the results were different: vaccinated individuals with confirmed Omicron or Delta breakthrough infections all showed the ability to neutralize all the tested variants, conferring higher protection. The biological samples used in the study were provided by Curative through its vaccine program.
“When it comes to other variants that might evolve in the future, we can’t predict exactly what would happen, but based on these results, I’d suspect that unvaccinated people who were infected with Omicron will have very little protection,” says Ott. “But on the contrary, vaccinated individuals are likely to be more broadly protected against future variants, especially if they had a breakthrough infection.”
“Our results may be useful not only to inform individuals’ decisions on vaccination, but also for the design of future COVID-19 vaccines that confer broad protection against many variants,” says Charles Chiu, MD, PhD, a professor of infectious diseases at UCSF and a co-senior author of the work.
For Media
Julie Langelier
Associate Director, Communications
415.734.5000
Email
About the Study
The paper “Limited Cross-Variant Immunity after Infection with the SARS-CoV-2 Omicron Variant Without Vaccination” was published in the journal Nature on May 17, 2022.
Other authors are Abdullah Syed, Camille Simoneau, Alison Ciling, Mir Khalid, Bharath Sreekumar, Pei-Yi Chen, Renuka Kumar, Mauricio Montano, Ronne Gascon, Frank Soveg, Ashley George, and Warner Greene of Gladstone; Noah Brazer, Prachi Saldhi, Miguel Garcia-Knight, Alicia Sotomayor-Gonzalez, Venice Servillita, Amelia Gliwa, Jenny Nguyen, Xiaohui Fang, Mazharul Maishan, Michael Matthay, and Raul Andino of UCSF; and Ines Silva, Bilal Milbes, Noah Kojima, Victoria Hess, Maria Shacreaw, Lauren Lopez, Matthew Brobeck, Fred Turner, and Lee Spraggon of Curative, Inc.
The work was supported by the National Institutes of Health (grants F31 AI164671-01, U54HL147127 and R21AI59666), the Natural Sciences and Engineering Research Council of Canada (PDF-533021-2019), the Roddenberry Foundation, Pamela and Edward Taft, the Howard Hughes Medical Institute, the Van Auken Private Foundation, David Henke, Emergent Ventures at the Mercatus Center (Fast Grants #2164 and #2208), George Mason University, the Innovative Genomics Institute, the US Centers for Disease Control and Prevention (75D30121C10991), Abbott Laboratories, and the Sandler Program for Breakthrough Biomedical Research at UCSF.
About Gladstone Institutes
Gladstone Institutes is an independent, nonprofit life science research organization that uses visionary science and technology to overcome disease. Established in 1979, it is located in the epicenter of biomedical and technological innovation, in the Mission Bay neighborhood of San Francisco. Gladstone has created a research model that disrupts how science is done, funds big ideas, and attracts the brightest minds.
Support Our COVID-19 Research Efforts
Gladstone scientists are moving quickly to respond to the coronavirus outbreak. Help us end this pandemic.
A Gene That Keeps Stem Cells From Losing Their Way
A Gene That Keeps Stem Cells From Losing Their Way
Scientists find a gene essential for keeping intestinal stem cells stable, offering insight into how tissues repair themselves.
News Release Research (Publication) Yamanaka Lab Stem Cells/iPSCsGladstone Recognized in Inaugural Cure Innovation Index for Translating Science Into Real-World Health Impact
Gladstone Recognized in Inaugural Cure Innovation Index for Translating Science Into Real-World Health Impact
Gladstone ranks 15th among 60 institutes and centers nationally for its ability to turn discoveries into measurable health outcomes.
Institutional News News ReleaseAI Discovery Reveals DNA Isn’t Locked Away in Cells After All
AI Discovery Reveals DNA Isn’t Locked Away in Cells After All
A new study upends long-held view about how DNA is stored, explaining how subtle shifts in gene activity contribute to cancer, aging, and other complex diseases.
News Release Research (Publication) Cancer Data Science and Biotechnology Ramani Lab Aging AI






