Gladstone NOW: The Campaign Join Us on the Journey✕
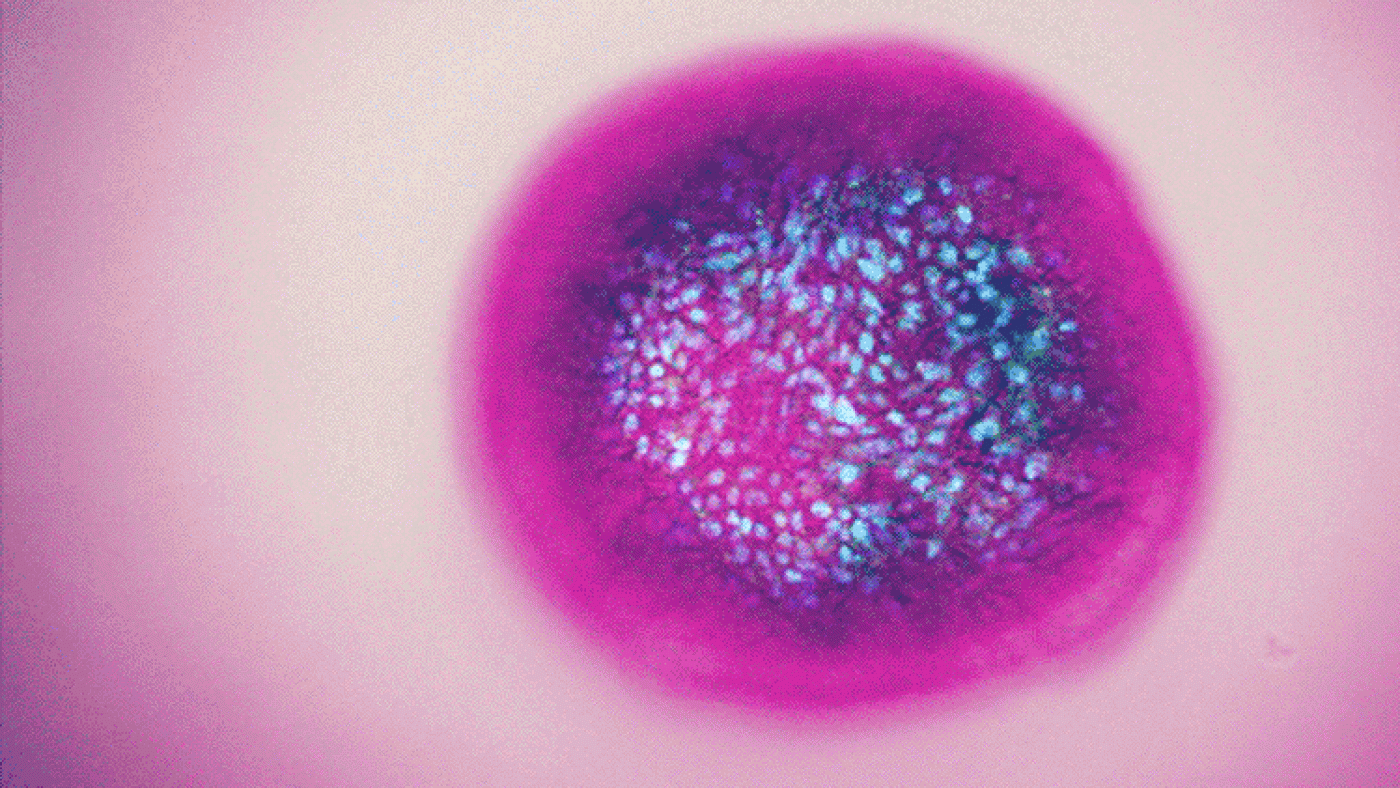
Liver organoids created from differentiated stem cells are helping Gladstone scientists study diseases that affect the liver, moving them one step closer to cures. [Image: Nathan Meyers, Gladstone Institutes]
Despite major advances made in diagnostic and imaging technology, there are still secrets of the human body that elude scientists. Researchers cannot learn the origins of memory loss by looking at the brain scan of an Alzheimer patient, and an electrocardiogram will not tell them why an arrhythmic heart has trouble beating. Instead, scientists have developed sophisticated models of disease in which to conduct experiments that would otherwise be impossible or unethical in humans.
Different types of model systems allow researchers to ask different types of questions. Models improve understanding of a disease, and they can be used to test potential treatments. At the Gladstone Institutes, scientists use animals, micro-organs, and stem cells to gain essential insights into deadly viruses, neurodegenerative disorders, and organ failure. These models enable the researchers to study human health and disease and to test potentially life-saving therapies.
Animals Are Essential for Viral Research
In many cases, scientists use a small animal, such as a rat or a mouse, to probe the origins and manifestations of diseases. Taking this approach, Gladstone Assistant Investigator Shomyseh Sanjabi, PhD, studies the sexual transmission of viruses in mice. She has obtained valuable information about how the mucosal immune system—the first line of defense against viral sexual transmission—springs into action against foreign invaders, and how the system can be defeated.
“It often takes days or even weeks to detect that a person has been infected with a virus, so it is impossible for us to study the first moments of viral transmission in humans,” Sanjabi explained. “Animal models are essential for us to understand the initial immune response to infection so that we can develop vaccines and treatments to stop transmission.”
One of the most important aspects of the body’s immune system is identifying self from other. If the immune system is too stringent, it can end up attacking the body’s own cells, resulting in auto-immune diseases, such as type I diabetes or inflammatory bowel disease. However, if it is too lax, pathogens can enter the body and go undetected. This balance is particularly delicate in the vaginal and rectal cavities, which are more amenable to foreign elements, such as sperm or healthy bacteria. Sanjabi thinks that viruses take advantage of these loopholes in the mucosal immune system to spread infection.
Sanjabi’s lab discovered that the vaginal immune system is suppressed in response to RNA viruses, including the Zika virus. A dampened and delayed immune response allows the virus to take hold and replicate in the vaginal cavity, where it can remain undetected for weeks. A serious potential risk during this time is for the virus to spread to the uterus, where it could infect a fetus. However, once the invader attempts to travel to the rest of the body, the central immune system detects the virus and eliminates it from the body.
Organoids Open Doors to New Discoveries
While mice can serve as good proxies for human immune responses, there are some diseases that are unique to people and cannot be modeled in animals. Gladstone Senior Investigators Melanie Ott, MD, PhD, and Todd McDevitt, PhD, are combining their expertise in virology and tissue engineering to study elusive viral infections in the newest type of disease model: organoids.
Using cells taken from liver biopsies, Ott and McDevitt are creating liver organoids—miniature, simplistic organs that consist of a few thousand cells—to study hepatitis B and hepatitis C.
“Both viruses are strictly human pathogens, so we cannot use animal models to study them,” Ott explained. “And while important discoveries about the viruses have been made using human liver cells, many aspects of the virus life cycle cannot be learned from individual cells. Organoids allow us to study the spread of the virus in a more realistic setting.”
Livers contain adult stem cells that divide naturally and have the capacity to turn into several different types of cells. Using stem cell and engineering tricks, the researchers coax the stem cells to mature into liver cells and grow together into large pieces of three-dimensional tissue.
“Right now, we can grow mini livers that resemble the native tissue in a dish using natural processes of development,” said McDevitt. “Our next step is to enrich the organoids with other types of cells to create even more realistic models of health and disease.”
With the liver organoids, Ott and McDevitt hope to gain a deeper understanding of how the viruses replicate, how they damage the liver, and how the immune system responds to them. Their ultimate goal is to find new approaches to develop a vaccine for hepatitis C and a better treatment for hepatitis B.
Stem Cells Facilitate Personalized Medicine
In the laboratory of Director Steven Finkbeiner, MD, PhD, scientists research neurodegenerative diseases using neurons developed from stem cells that were created from patients’ skin cells. This approach allows the scientists to study brain cells that have the same DNA as the patient—something that would be virtually impossible were it not for induced pluripotent stem cell technology.
The scientists use these neurons to research the onset and progression of Huntington’s disease, Parkinson’s disease, and amyloid lateral sclerosis. They look for changes in the cells that might explain how a gene mutation or disruption of a single protein can lead to neuron dysfunction and death. Ultimately, the researchers hope to identify cellular characteristics that can be directly related to the patient’s symptoms.
“I believe that there are predictive relationships between the models that we build in the lab with induced pluripotent stem cells and the patients themselves,” Finkbeiner said. “We can measure hundreds of different traits in the cells and track how those traits change over time. At the same time, we can deeply characterize patients from whom those cells were derived. Then, we can determine if any of those traits could predict a patient’s clinical progression.”
The ability to predict a patient’s disease progression would be especially valuable when considering treatment options. That is because two patients diagnosed with the same disorder can have different root causes for the disease. For example, Parkinson’s disease can arise from a mutation in one of several genes, or it can develop spontaneously. The distinct disease origins can result in different symptoms, and, more importantly, may require different courses of treatment.
Many clinical trials are not sensitive enough to distinguish between subsets of patients with the same condition, and some scientists, including Finkbeiner, think this may be why so many clinical trials for neurodegenerative diseases fail. However, if the researchers could first conduct a “clinical trial in a dish” on a patient’s cells, they would have a better understanding of that person’s disease subset and the likelihood of whether or not a given treatment would be successful.
Finkbeiner says that achieving this goal would allow researchers and physicians to conduct better clinical trials and could potentially lead to a personalized medicine approach to treating neurodegenerative diseases.
Better Models Lead to Better Treatments
The development of novel research methods and technologies at Gladstone and elsewhere continues to open doors to innovative experiments and new discoveries. By having a diverse array of models available, scientists can study virtually every organ system in the body and make important breakthroughs about health and disease. Ultimately, knowledge gained and potential treatments identified through research in mice, cells, and organoids can be applied to people, providing patients with new therapies.
How AI Is Pinpointing the Genetic Cause of Disease
How AI Is Pinpointing the Genetic Cause of Disease
Scientists at Gladstone are combining AI models with cutting-edge lab tools to finally make sense of the human genome.
AI Srivastava Lab Pollard Lab Engelhardt Lab Theodoris Lab Shipman Lab Ramani Lab Finkbeiner Lab Cardiovascular Disease Neurological DiseaseHow AI Is Accelerating Life-Saving Discovery
How AI Is Accelerating Life-Saving Discovery
Gladstone scientists are developing new AI tools that promise to revolutionize how science is done and lead to new treatments for the most devastating diseases.
AI Srivastava Lab Pollard Lab Engelhardt Lab Theodoris Lab Shipman Lab Ramani Lab Finkbeiner Lab Cardiovascular Disease Neurological DiseaseMeet Gladstone: Shijie Wang
Meet Gladstone: Shijie Wang
Shijie Wang, a postdoctoral scholar in Steve Finkbeiner’s lab, uses artificial intelligence, robotics, and stem cell technologies to uncover how brain cells die in neurodegenerative diseases like Alzheimer’s and Parkinson’s.
Profile Neurological Disease Finkbeiner Lab AI Robotic Microscopy



