Gladstone NOW: The Campaign Join Us on the Journey✕

Jorge Palop (left) and Katerina Akassoglou (right) received an NIH grant to study the link between neurovascular dysfunction and cognitive decline in Alzheimer’s disease.
Alzheimer’s disease is a progressive neurodegenerative disease that causes loss of memory. Despite decades of research into how the disease causes brain cells to die, there is no cure.
In the last several years, a string of clinical trials for Alzheimer’s disease therapies have failed. These failures suggest that scientists need to take a fresh look at what drives Alzheimer’s disease to design new approaches.
Researchers at Gladstone Institutes are doing just that. A team led by Senior Investigator Katerina Akassoglou, PhD, was recently awarded over $4.5 million from the National Institutes of Health (NIH) to study how dysfunction of blood vessels in the brain (the neurovascular system) can lead to neurodegeneration.
“There is an emerging interest in the contribution of vascular dysfunction to Alzheimer’s disease. We know that in Alzheimer’s disease, changes to blood vessels in the brain occur really early, and they correlate and often precede dementia,” explained Akassoglou, who is also a professor of neurology at UC San Francisco.
However, whether and how this vascular dysfunction causes cognitive decline remains a mystery.
Akassoglou and her team previously showed that one consequence of vascular dysfunction is that proteins normally found in the blood can leak into the brain due to a compromised blood-brain barrier. Specifically, their research pinpointed a damaging effect of fibrinogen, a blood protein important for clotting, that is deposited in the brains of Alzheimer’s disease patients. When fibrinogen escapes the blood vessels and enters the brain, it can activate the brain’s immune cells and trigger them to destroy important connections between neurons, hastening the loss of synapses and entire nerve cells.
Another finding that has emerged from recent research is that the activity of neurons—the rate and rhythm with which they fire—is altered in the brains of Alzheimer’s disease patients. Research from Gladstone investigators Jorge Palop, PhD, and Lennart Mucke, MD, has shown that these changes to neuronal activity can occur decades before the onset of Alzheimer’s symptoms. Alzheimer’s-associated changes in neuronal activity also include seizures.
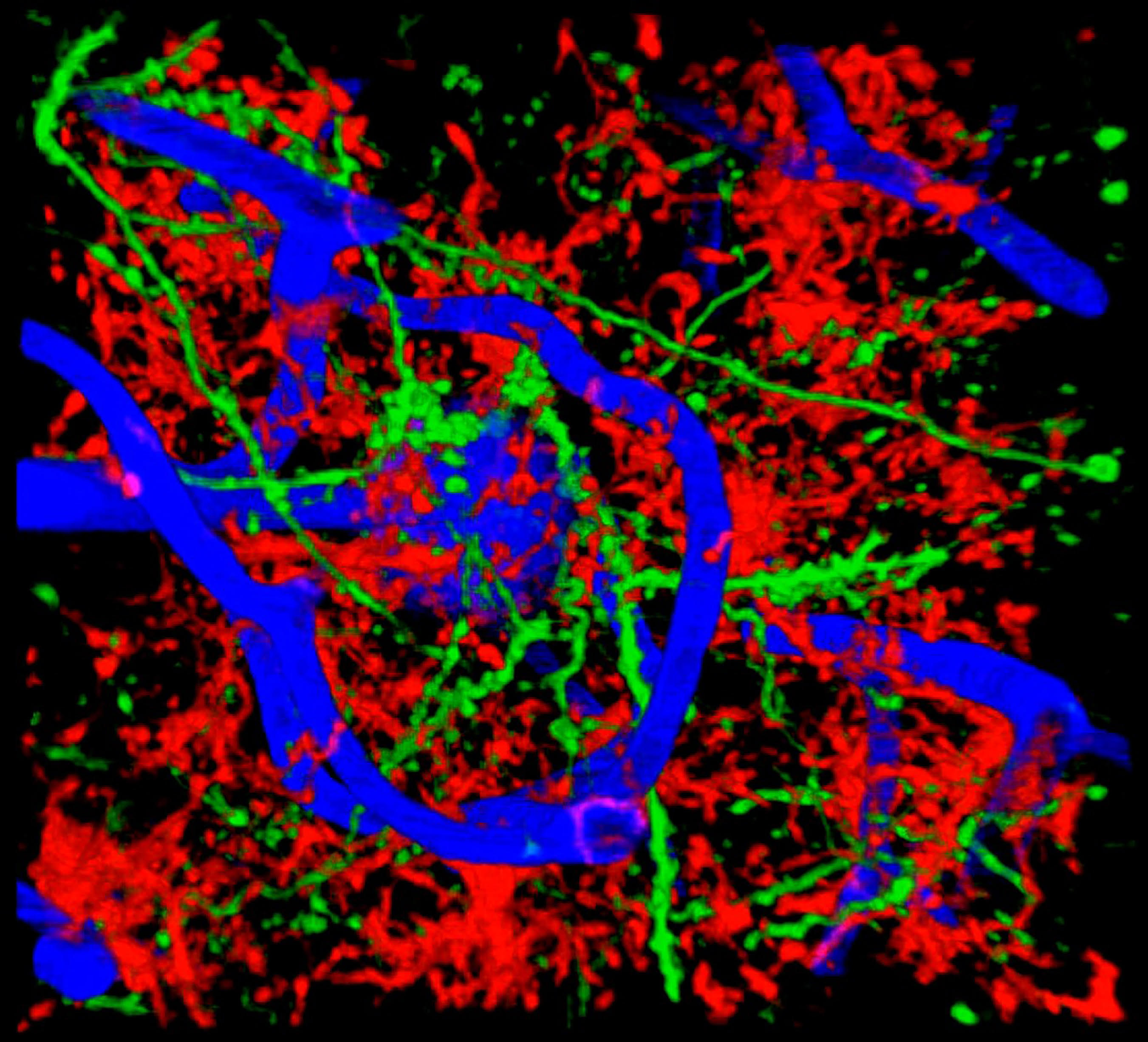
Live imaging of blood vessels, microglia, and neurites. Image credit: Mario Merlini, Akassoglou Lab
This new NIH grant will support Akassoglou, Palop, and a third collaborator, Mark Ellisman, at UC San Diego, as they join forces to investigate whether these two aspects of Alzheimer’s disease—vascular dysfunction and excessive neuronal activity—are related, and whether blocking one can prevent the other.
“How dysfunction of the vascular and immune systems alters ongoing neuronal activity is a critical question in the study of Alzheimer’s disease,” said Palop. “We can record brain electrical activity in a mouse for weeks at a time and observe how vascular damage affects neuronal function.”
“We know that the vascular alterations and blood leaks are present in Alzheimer’s disease, but we do not know how they affect neuronal activity.” said Akassoglou. “Connecting the dots between blood leaks, the brain’s immune cells, and neuronal activity could change the way we think how cognitive decline develops and be the cornerstone for developing new treatments.”
The project builds on a long-running collaboration between Akassoglou and Ellisman, a distinguished professor at UC San Diego School of Medicine and an expert in multiscale microscopy. Ellisman is the director of the National Center for Microscopy and Imaging Research, and is a pioneer in the development of 3-dimensional light and electron microscopy applied to the study of the structure and function of normal and abnormal nervous systems.
By combining state-of-the-art imaging techniques with recordings of neuronal activity, the researchers hope to discover the series of events that lead to cognitive decline in Alzheimer’s disease. They also hope to identify new therapeutic targets that might prevent this process.
“It’s critical to understand the relationships between vascular, neuronal, and immune dysfunctions,” said Lennart Mucke, director of the Gladstone Institute of Neurological Disease. “This work and related studies by other groups in our institute will be invaluable in the design of novel therapies for Alzheimer’s disease and a range of other impactful brain disorders.”
For Media
Julie Langelier
Associate Director, Communications
415.734.5000
Email
About Gladstone Institutes
Gladstone Institutes is an independent, nonprofit life science research organization that uses visionary science and technology to overcome disease. Established in 1979, it is located in the epicenter of biomedical and technological innovation, in the Mission Bay neighborhood of San Francisco. Gladstone has created a research model that disrupts how science is done, funds big ideas, and attracts the brightest minds.
Support Discovery Science
Your gift to Gladstone will allow our researchers to pursue high-quality science, focus on disease, and train the next generation of scientific thought leaders.
Gladstone Launches Center for PhAIge Therapy to Harness AI in the Fight Against Drug-Resistant Infections
Gladstone Launches Center for PhAIge Therapy to Harness AI in the Fight Against Drug-Resistant Infections
The center, funded by an NIH grant, will become one of three national centers dedicated to accelerating the development of phage therapy.
News Release Grants Data Science and Biotechnology Infectious Disease Shipman Lab Pollard Lab Ott Lab Silas Lab AIGladstone Investigator Andrew Yang Receives 2026 Sobrato Prize in Neuroscience
Gladstone Investigator Andrew Yang Receives 2026 Sobrato Prize in Neuroscience
The prize honors scientists whose breakthroughs in brain research have high potential for patient impact.
Philanthropy Awards News Release Alzheimer’s Disease Neurological Disease Yang LabMapping How the Brain Takes Out Its Trash
Mapping How the Brain Takes Out Its Trash
Gladstone scientists developed a new tool to trace how the brain clears “waste,” uncovering surprising new biology about how it keeps itself healthy.
News Release Research (Publication) Alzheimer’s Disease Neurological Disease Yang Lab



